Toujeo is proven to consistently reduce A1C
In clinical studies, Toujeo provided stable blood sugar control all day and all night
In an early study of patients on Toujeo, blood sugar levels stayed within the American Diabetes Association (ADA) target zone for more than 24 hours.*
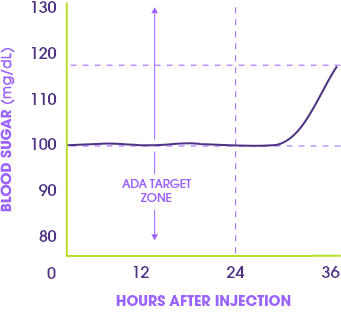
This study compared Toujeo (Gla-300) with the standard version (Gla-100) in people with type 1 diabetes. Toujeo released insulin more steadily and lasted longer in the body than Gla-100. As a result, it kept blood sugar under control lasting over 24 hours and about 5 hours longer than Gla-100. These findings suggest Toujeo may help people maintain smoother, longer-lasting blood sugar control.

Toujeo should be taken at the same time every day, once a day.
Results based on a study of 30 patients with type 1 diabetes; study size is typical for this type of study. May not apply in everyday use. Target zone: 80-130 mg/dL.
*American Diabetes Association. Diabetes Care. 2025;45(Suppl1):S83-S96.

Most common and serious side effects:
For all insulins, including Toujeo, the most common side effect is hypoglycemia. Ask your doctor about the signs and symptoms of hypoglycemia, how to monitor your blood sugar, and what to do if you have a hypoglycemic event.
Other common side effects (<= 5%) include allergic reactions, injection site reaction, pitted or thickened skin, itchiness, rash, swelling or redness, and weight gain.
These are not all the possible side effects of Toujeo. Talk with your doctor about possible side effects.
For more detailed information, see the full Important Safety Information and full Prescribing Information.

Toujeo SoloStar and Toujeo Max SoloStar are longer-acting insulin pens
The body releases insulin throughout the day to keep your blood sugar stable. Insulins like Toujeo are meant to do essentially the same. Taken once a day, long-acting insulin simulates how your body controls blood sugar.

Understanding Time in Range and Continuous Glucose Monitoring
Time in Range is the amount of time your blood sugar levels are within a healthy range. This is important because it helps your doctor know how well your treatment is working.
To accurately monitor Time in Range, consider a Continuous Glucose Monitoring (CGM) system. It’s a small sensor attached to your body that automatically tests your glucose every few minutes. In addition to measuring Time In Range, most CGM systems can alert you when your blood sugar levels are high or low. Ask your doctor if a CGM system can help you manage diabetes.

What more can you do to manage your diabetes?
Maintaining a healthy diet and exercising regularly can help you keep your A1C under control. Join TeamingUp to get tips on maintaining a healthy lifestyle.
.2023-06-29-20-47-56.png 252w)
Keeping Track of Your Blood Sugar
Writing down blood sugar numbers, dose, and activity can help your doctor keep a plan that manages your diabetes effectively. Download the blood sugar log to help and share it with your doctor.
What Insulin Does
Insulin is a naturally occurring hormone your pancreas produces that regulates blood sugar (glucose) in your body. It’s like a key that lets blood sugar into cells to use for energy.
About type 2 diabetes
Most people who have diabetes have type 2 diabetes. For these people, the pancreas does not produce enough insulin and/or the body can’t properly use the insulin it produces to keep their blood sugar controlled. You may hear healthcare professionals refer to it as insulin resistance.
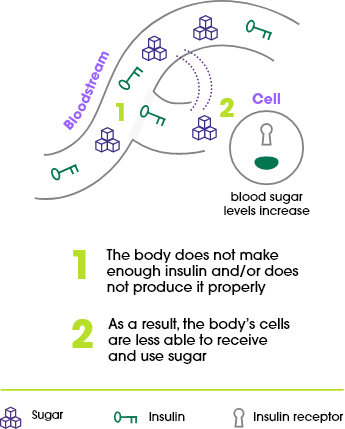
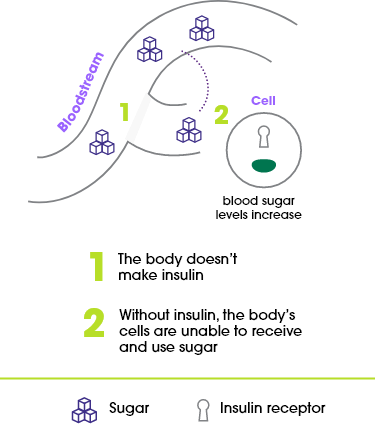
About type 1 diabetes
For people with type 1 diabetes, the body does not produce insulin at all. It’s managed with insulin and other treatments.

Pay no more than $35† per month for Toujeo Max
Whether you have commercial insurance or not, or prefer to pay cash outside of your prescription drug benefit, we have an offer for you.

What do people think of Toujeo Max?
See what others have said about their experience with Toujeo Max and managing diabetes.
What is Toujeo U-300 (insulin glargine) injection 300 Units/mL?
Prescription Toujeo is a long-acting man-made insulin used to control high blood sugar in adults and children who are 6 years of age and older with diabetes mellitus.
- Toujeo is not for the treatment of diabetic ketoacidosis
- It is not known if Toujeo is safe and effective in children under 6 years of age
Important Safety Information
Important Safety Information
Do not use Toujeo if you have low blood sugar or if you are allergic to insulin or any of the ingredients in Toujeo.
Do not share your pen(s) with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
Before starting Toujeo, tell your doctor about all your medical conditions, including if you have liver or kidney problems, if you are pregnant or planning to become pregnant, or if you are breastfeeding or planning to breastfeed.
Change (rotate) your injection sites within the area you chose with each dose to reduce your risk of getting pitted or thickened skin (lipodystrophy) and skin with lumps (localized cutaneous amyloidosis) at the injection sites. Do not use the same spot for each injection or inject where the skin is pitted, thickened, lumpy, tender, bruised, scaly, hard, scarred, or damaged.
Heart failure can occur if you are taking insulin together with pills called TZDs (thiazolidinediones), even if you have never had heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Toujeo. Your treatment with TZDs and Toujeo may need to be changed or stopped by your doctor if you have new or worsening heart failure. Tell your doctor if you have any new or worsening symptoms including:
- Shortness of breath
- Sudden weight gain
- Swelling of your ankles or feet
Tell your doctor about all the medications you take, including OTC medicines, vitamins, and supplements, and herbal supplements.
Toujeo should be taken at the same time once a day. Test your blood sugar levels daily while using any insulin. Do not change your dose or type of insulin without talking to your doctor. Verify you have the correct insulin before each injection. Do NOT use a syringe to remove Toujeo from your pen. Your dose for Toujeo may be different from other insulins you have taken. Any change of insulin should be made cautiously and only under medical supervision.
Do NOT dilute or mix Toujeo with any other insulin or solution. It will not work as intended and you may lose blood sugar control, which could be serious. Use Toujeo only if the solution is clear and colorless with no particles visible.
While using Toujeo, do not drive or operate heavy machinery until you know how Toujeo affects you. Don’t drink alcohol or use other medicines that contain alcohol.
The most common side effect of Toujeo is low blood sugar (hypoglycemia), which may be serious and life-threatening. Severe hypoglycemia may cause harm to your heart or brain. Symptoms of serious low blood sugar may include shaking, sweating, fast heartbeat, and blurred vision. The long-acting effect of Toujeo may delay recovery from low blood sugar compared to shorter-acting insulins.
Toujeo may cause severe allergic reactions that can lead to death. Get medical help right away if you have:
- A rash over your whole body
- Shortness of breath
- Swelling of your face, tongue, or throat
- Extreme drowsiness, dizziness, or confusion
- Trouble breathing
- Fast heartbeat
- Sweating
Toujeo may have additional side effects including swelling, weight gain, low potassium, and injection site reactions which may include change in fat tissue, skin thickening, redness, swelling, and itching.
Toujeo SoloStar and Toujeo Max SoloStar are single-patient-use prefilled insulin pens. It is important to perform a safety test when using a new pen for the first time. Talk to your doctor about proper injection technique and follow instructions in the Instruction Leaflet that comes with your Toujeo SoloStar or Toujeo Max SoloStar pen.
†Eligibility Restrictions & Offer Terms:
Sanofi Insulins Co-pay Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is not valid for prescriptions covered by or submitted for reimbursement, in whole or in part, under Medicare, Medicaid, VA, DOD, TRICARE, similar federal or state programs, including any state pharmaceutical programs. If you have an Affordable Care (Health Care Exchange) plan, you may still be qualified to receive and use this savings card. Please note: the Federal Employees Health Benefits (FEHB) Program is not a federal or state government health care program for purposes of the savings program. Void where prohibited by law. For the duration of the program, eligible commercially insured patients pay no more than $35 per 30-day supply, up to 10 packs per fill; Offer valid for one fill every 30 days. Savings may vary depending on patients’ out-of-pocket costs. The Sanofi Insulins Co-pay Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Sanofi Insulins Co-pay Savings Program at (866) 255-8661 (866) 255-8661 (9:00 am-7:00 pm EST, Monday-Friday).
Insulins Valyou Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is only valid for those who are uninsured or those who are insured by a prescription plan but are not using such insurance and will be paying the full retail price for the medication. Void where prohibited by law. The Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. You may not submit claims for reimbursement to any third-party payor, including any government healthcare plan (e.g., Medicare, Medicaid, DOD, VA, TRICARE) or similar federal or state programs for Sanofi Insulin prescriptions when using this Program. You may not seek to have your out-of-pocket costs or the full retail price of the Sanofi Insulin count toward your deductible, true-out-of-pocket (TrOOP), maximum out-of-pocket (MOOP), or any other out-of-pocket caps associated with any insurance coverage. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Insulins Valyou Savings Program at (833) 813-0190 (833) 813-0190 (9:00 am-7:00 pm EST, Monday-Friday).
Important Safety Information
Do not use Toujeo if you have low blood sugar or if you are allergic to insulin or any of the ingredients in Toujeo.
Do not share your pen(s) with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
Before starting Toujeo, tell your doctor about all your medical conditions, including if you have liver or kidney problems, if you are pregnant or planning to become pregnant, or if you are breastfeeding or planning to breastfeed.
Change (rotate) your injection sites within the area you chose with each dose to reduce your risk of getting pitted or thickened skin (lipodystrophy) and skin with lumps (localized cutaneous amyloidosis) at the injection sites. Do not use the same spot for each injection or inject where the skin is pitted, thickened, lumpy, tender, bruised, scaly, hard, scarred, or damaged.
Heart failure can occur if you are taking insulin together with pills called TZDs (thiazolidinediones), even if you have never had heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Toujeo. Your treatment with TZDs and Toujeo may need to be changed or stopped by your doctor if you have new or worsening heart failure. Tell your doctor if you have any new or worsening symptoms including:
- Shortness of breath
- Sudden weight gain
- Swelling of your ankles or feet
Tell your doctor about all the medications you take, including OTC medicines, vitamins, and supplements, and herbal supplements.
Toujeo should be taken at the same time once a day. Test your blood sugar levels daily while using any insulin. Do not change your dose or type of insulin without talking to your doctor. Verify you have the correct insulin before each injection. Do NOT use a syringe to remove Toujeo from your pen. Your dose for Toujeo may be different from other insulins you have taken. Any change of insulin should be made cautiously and only under medical supervision.
Do NOT dilute or mix Toujeo with any other insulin or solution. It will not work as intended and you may lose blood sugar control, which could be serious. Use Toujeo only if the solution is clear and colorless with no particles visible.
While using Toujeo, do not drive or operate heavy machinery until you know how Toujeo affects you. Don’t drink alcohol or use other medicines that contain alcohol.
The most common side effect of Toujeo is low blood sugar (hypoglycemia), which may be serious and life-threatening. Severe hypoglycemia may cause harm to your heart or brain. Symptoms of serious low blood sugar may include shaking, sweating, fast heartbeat, and blurred vision. The long-acting effect of Toujeo may delay recovery from low blood sugar compared to shorter-acting insulins.
Toujeo may cause severe allergic reactions that can lead to death. Get medical help right away if you have:
- A rash over your whole body
- Shortness of breath
- Swelling of your face, tongue, or throat
- Extreme drowsiness, dizziness, or confusion
- Trouble breathing
- Fast heartbeat
- Sweating
Toujeo may have additional side effects including swelling, weight gain, low potassium, and injection site reactions which may include change in fat tissue, skin thickening, redness, swelling, and itching.
Toujeo SoloStar and Toujeo Max SoloStar are single-patient-use prefilled insulin pens. It is important to perform a safety test when using a new pen for the first time. Talk to your doctor about proper injection technique and follow instructions in the Instruction Leaflet that comes with your Toujeo SoloStar or Toujeo Max SoloStar pen.
†Eligibility Restrictions & Offer Terms:
Sanofi Insulins Co-pay Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is not valid for prescriptions covered by or submitted for reimbursement, in whole or in part, under Medicare, Medicaid, VA, DOD, TRICARE, similar federal or state programs, including any state pharmaceutical programs. If you have an Affordable Care (Health Care Exchange) plan, you may still be qualified to receive and use this savings card. Please note: the Federal Employees Health Benefits (FEHB) Program is not a federal or state government health care program for purposes of the savings program. Void where prohibited by law. For the duration of the program, eligible commercially insured patients pay no more than $35 per 30-day supply, up to 10 packs per fill; Offer valid for one fill every 30 days. Savings may vary depending on patients’ out-of-pocket costs. The Sanofi Insulins Co-pay Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Sanofi Insulins Co-pay Savings Program at (866) 255-8661 (866) 255-8661 (9:00 am-7:00 pm EST, Monday-Friday).
Insulins Valyou Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is only valid for those who are uninsured or those who are insured by a prescription plan but are not using such insurance and will be paying the full retail price for the medication. Void where prohibited by law. The Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. You may not submit claims for reimbursement to any third-party payor, including any government healthcare plan (e.g., Medicare, Medicaid, DOD, VA, TRICARE) or similar federal or state programs for Sanofi Insulin prescriptions when using this Program. You may not seek to have your out-of-pocket costs or the full retail price of the Sanofi Insulin count toward your deductible, true-out-of-pocket (TrOOP), maximum out-of-pocket (MOOP), or any other out-of-pocket caps associated with any insurance coverage. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Insulins Valyou Savings Program at (833) 813-0190 (833) 813-0190 (9:00 am-7:00 pm EST, Monday-Friday).

